 This method helps us in describing the formation of ionic compounds from different elements. Observe and analyze energies in a reaction. The method also exhibits several properties such as electron affinity, ionization energy, sublimation energy, the heat of formation and dissociation energy. It is primarily used to calculate the lattice energy which is a measure of the strength of ionic bonds that exist in an ionic compound involved in a chemical reaction. This methodology was introduced in the year 1919 by German scientists Fritz Haber and Max Born. It further enables us to understand the overall reaction process through a series of steps involved in it. This process describes the formation of ionic compounds from different elements. However, on the curse side of the Haber process, we have several issues including serious imbalances to the nitrogen cycle, high fossil fuel energy inputs, negative effects on soil organisms and soil organic matter, excess runoff that cause ocean dead zones, and it's a major component of weapons (including all those roadside bombs).Īn introduction to the brilliant Fritz Haber, a great mind who is considered " the father chemical warfare.Born Haber process or more commonly known as Born Haber cycle is a method in use that lets us observe and analyze energies in a reaction. It's safe to say that the Haber process has led to some amazing advancements in scientific history. Notably, the rise of the Haber industrial process led to the "Nitrate Crisis" in Chile when the natural nitrate mines were no longer profitable and were closed, leaving a large unemployed Chilean population behind. That fertilizer is responsible for sustaining one-third of the Earth's population, but results in various deleterious environmental consequences. 3–5% of the world's natural gas production is consumed in the Haber process (~1–2% of the world's annual energy supply). The Haber process now produces 500 million short tons (454 million tonnes) of nitrogen fertilizer per year, mostly in the form of anhydrous ammonia, ammonium nitrate, and urea. Within a year after the German introduction of dichlordiethylsulfide, the Allies backed by superior productive capacity and a more efficient manufacturing method, were retaliating with deadly attacks. He recommended the use of mustard gas only if Germany could win within a year otherwise not. The top command didn't heed him on another occasion, which they later regretted. As a surprise weapon, it might have shattered the Allies, had the generals listened to him. Haber knew poison gas was at first a morale weapon, a device against men's minds accustomed to the usual. Until early 1915, the Powers were locked in trench warfare, and poison gas drove the armies from dugouts. He knew its tactical value-to lead to movement in battle and pave the way to victory for one side or the other. Fritz Haber correctly analyzed the functions of poison gas. Every detail of chemical defense and offense, supply and research came under his supervision.  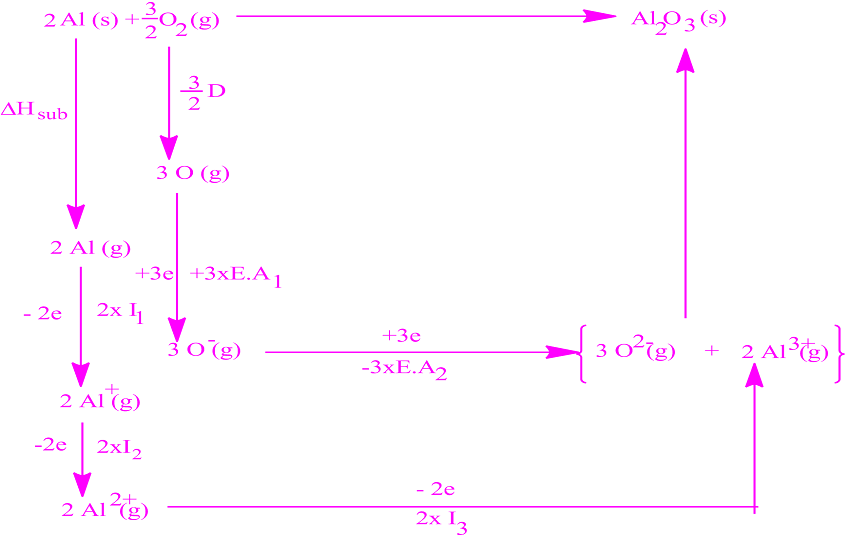 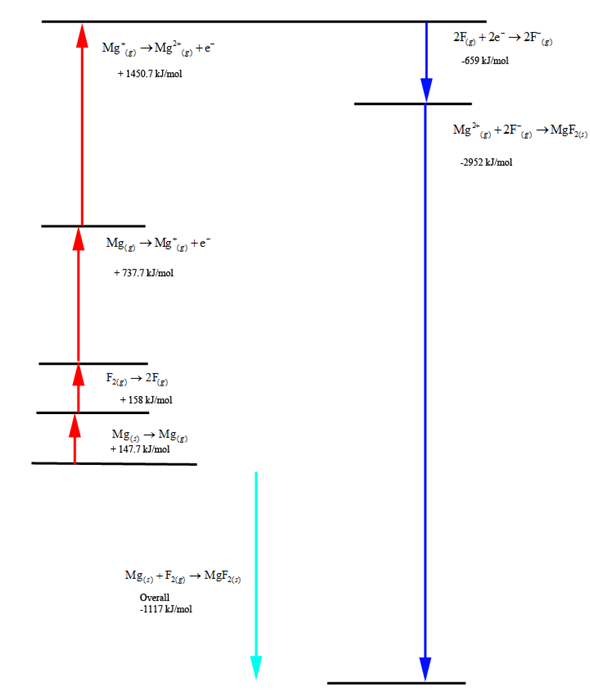
He directed poison gas offense and defense activities, and later had charge of all chemical procurement and planning. Within a short time, he was promoted directly to captain, a very uncommon feat in the Wehrmacht. "He said, if you want to win the war, then please, wage chemical warfare with conviction."įritz Haber started his military career as a sergeant-major in the artillery. "Haber actually insisted on this," says biographer Margit Szöllösi-Janze. His idea: to use poison gas to destroy enemy trenches. When the war turned into a stalemate, with both sides stuck in trenches, Haber tried to break the deadlock with chemistry. Like Einstein, Haber was Jewish and German unlike Einstein, he converted to Christianity and was a German patriot. For more information, read Between Genius and Genocide: The Tragedy of Fritz Haber, Father of Chemical Warfare by Daniel Charles. The research results show the ambivalence of his scientific activity: on the one hand, development of ammonia synthesis for the manufacture of explosives and of a technical process for the industrial manufacture and use of poison gas in warfare but on the other hand, development of an industrial process without which the food supply for today's world population would be greatly diminished. Haber received much criticism for his involvement in the development of chemical weapons in pre-World War II Germany, both from contemporaries and from modern-day scientists. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
